| Artificial sponge-like materials, combining the elastic and mechanical properties of a sponge with chemical functionality, have a wide range of applications, from use as scaffolds for the growth of biological tissue to mechanical actuators and catalysis. Electrically conductive sponges, or elastomeric conductors, expand the range of possible applications even further, but to date have involved complex preparation methods such as chemical vapor deposition of carbon nanotubes or the costly production of carbon nanotube gels. | |
| Shu-Hong Yu and colleagues from the University of Science and Technology of China, Hefei, have now devised a much simpler approach for producing elastomeric conductors using carbonized silver–polymer nanocables ("Macroscale Elastomeric Conductors Generated from Hydrothermally Synthesized Metal-Polymer Hybrid Nanocable Sponges"). | |
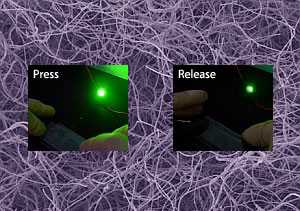 | |
| Compression and decompresson changes the electrical resistance of the silver–carbon conductive sponge. (© 2011 Wiley-VCH) | |
| Using an inexpensive and scalable one-pot hydrothermal method, Yu and his colleagues grew silver-cross-linked poly(vinyl alcohol) coaxial nanocables into an interconnected network to form a sponge-like three-dimensional macrostructure. They then freeze-dried the sponges and heated them at high temperature, converting the threads into hybrid silver–carbon nanocables. The resulting materials are soft, flexible and elastic. At present, the conductivity of the silver–carbon sponge is lower than that of its carbon nanotube counterparts, but as Yu notes, "further optimization of the preparation procedures is expected to result in improved conductivity." | |
| By placing the silver-polymer sponge between two pieces of glass and connecting a light-emitting diode (LED), Yu and his colleagues were able to show how the relationship between compressive deformation and conductivity can be exploited to produce pressure sensor functionality (see image). Depressing the cover glass causes the electrical resistance to decrease and the LED to become brighter, and on releasing the pressure the LED quickly returns to its original luminosity. The researchers also demonstrated the possibility of creating nanocomposites by infiltrating the conductive sponge with another polymer. | |
| Yu and colleagues next plan to investigate the functionalization of these sponge-like silver–carbon nanocable networks so they can be used as bio-sensors for the detection of biological molecules such as enzymes, and as electrode materials in high-performance lithium batteries and flexible supercapacitors. The researchers expect that functionalization for such applications can be achieved by coating the silver–carbon nanocables with various metal oxides in order to introduce active sites onto the backbone of the conductive networks. |
Fuente: |



No comments:
Post a Comment